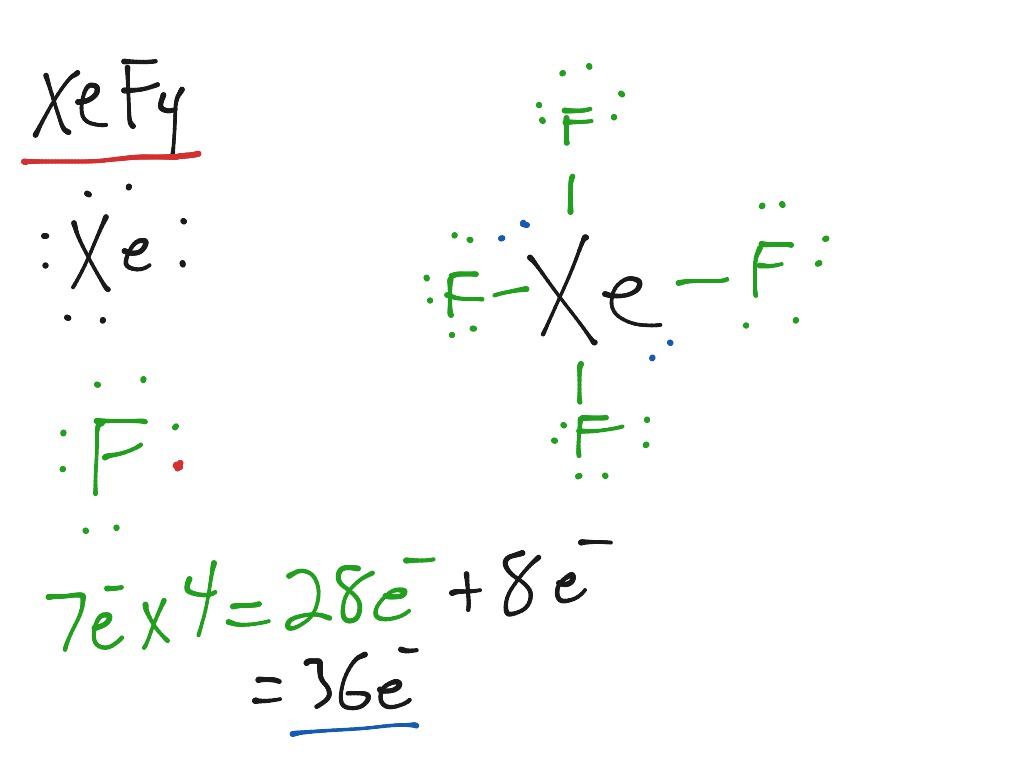
The four bonded atoms lie on the 2-D plane giving the compound a square planar shape. Two orbitals contain lone pairs of electrons on opposite sides of the central atom.The shape of the orbitals in a square planar is octahedral.The hybridisation of 3d orbitals with 4s and 4p orbitals is termed outer orbital complexes, as the principal quantum number of the orbitals except the d-orbitals is one unit higher.If they hybridise with the former group, they form inner orbital complexes, as the principal quantum number for all the orbitals is the same. Thus it makes the d-orbitals hybridise with both groups. The energy of 3d-orbitals of an atom is either similar to 3s and 3p or 4s and 4p.The hybridisation that is achieved by this shape is dsp2. It is a planar structure having one central atom bonded to 4 other atoms. Square planar is one of the geometries characterising a specific hybridisation of atoms.Some aspects are the number and position of lone pairs, type of substituent attached to the central atom, etc.But some factors affect the geometry and therefore alter the overall shape of the molecules. The hybrid orbitals have some characteristic geometry from the time of formation.The hybridisation of atomic orbitals forms a new set of orbitals called hybridised orbitals to attain better stability.The bonding pairs experience equal repulsion from both lone pairs, so the four bonds are split on a 2-D plane with 90 degrees of separation between each bonded pair. In a square planar-shaped molecule, the lone pairs are on opposite sides of the central atom of the molecule, with the four bonded neighbouring atoms all lying in one plane.The bond angle in square planar is 90 degrees.According to VSEPR Theory, a square planar molecular geometry corresponds to an AB4C2 molecule.The theory assumes that the molecule tries to achieve a geometry in which the repulsion between the electrons in the atom’s valence shell is minimum.of lone pairs surrounding the central atom. VSEPR theory is a model that helps us predict the geometrical structure of various molecular compounds with the help of the no. 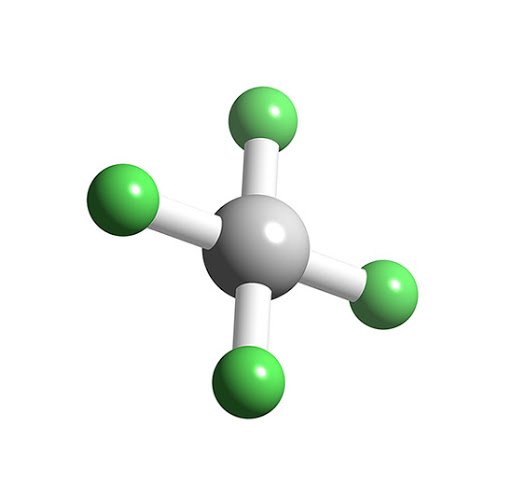
The XeF4 molecule comprises six equally spaced sp3d2 (or d2sp3) hybrid orbitals arranged at 90° angles. Xenon tetrafluoride (XeF4) is a molecular compound with a square planar structure. When there are four bonds and two lone pairs on the molecule’s central atom, this shape is Square Planar. The Square Planar molecular geometry tells us about the stereochemistry of various molecular compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
